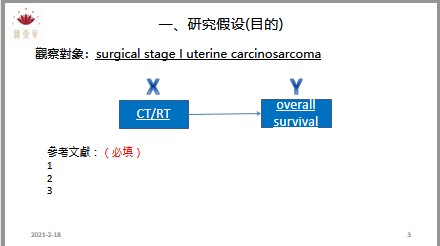
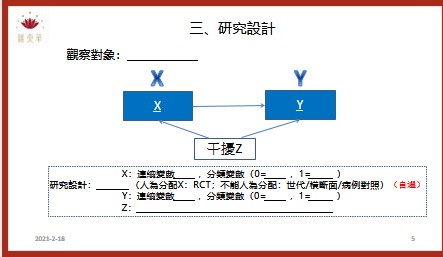
The effectiveness of topical chloramphenicol in preventing wound infection after minor dermatological surgery was evaluated. A randomised placebo controlled double blind superiority trial was performed. The intervention was a single application of topical chloramphenicol ointment applied to the sutured wound immediately after suturing. Participants were patients with high risk sutured wounds who had undergone minor surgery.1
The primary outcome was infection on the agreed day of removal of sutures or sooner if the patient re-presented with a perceived infection. The required sample size was based on a projected infection rate of 10% in the placebo group. The
smallest effect of clinical interest was an absolute decrease in the incidence of infection of 5%. To achieve this difference with a power in excess of 80% and a critical level of significance of 0.05, 473 patients were needed in each treatment group. In total, 972 patients were recruited and randomised to topical chloramphenicol ointment (n=488) or placebo (n=484).
The proportion of participants with an infection in the topical chloramphenicol group was statistically significantly lower than for placebo (6.6% v11.0%; difference −4.4%, 95% confidence interval −7.9% to −0.8%; P=0.010).
Which of the following statements, if any, are true?
a) The derived sample size was based on clinical significance
b) The derived sample size was based on statistical significance
c) It can be inferred that the reduction in infection rate for the intervention compared with placebo was clinically significant because it was statistically significant
 匯東華統計顧問有限公司
匯東華統計顧問有限公司 
![]()
![]()

![]()
![]()