 匯東華統計顧問有限公司
匯東華統計顧問有限公司
"匯東華-認真作好每件事"
~統計,不再是阻力,而是助力~
![]()
BMJ小小統計問題(24)
![]()
數據串接與清洗
數據是礦藏,數據清洗是挖出鑽石的第一步,尤其是巨量知識。數據清洗或串接執行過程需要細心與專注,且有可能會消耗許多時間和精力,就由我們來替各位處理掉這個大麻煩。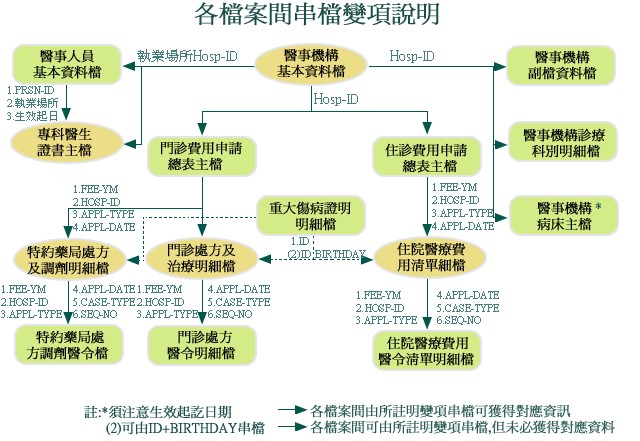
全民健保研究資料庫、國外大型資料庫資料非常齊全,種類多,需要串接與清洗,進行正規化後才能更進一步進行資料探勘與統計分析。

Fig1.同一個Project資料散落在不同tables,無法使用
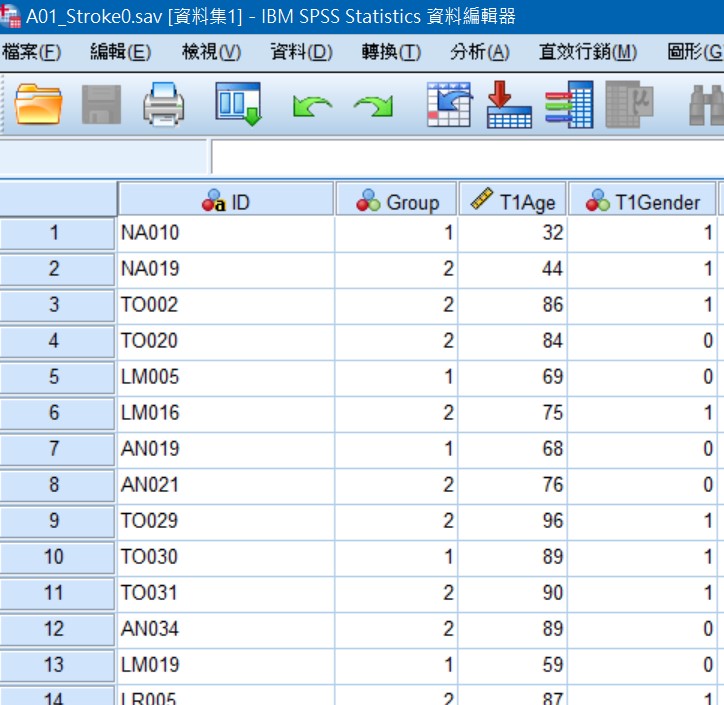
Fig2.整併與清理為可分析的table
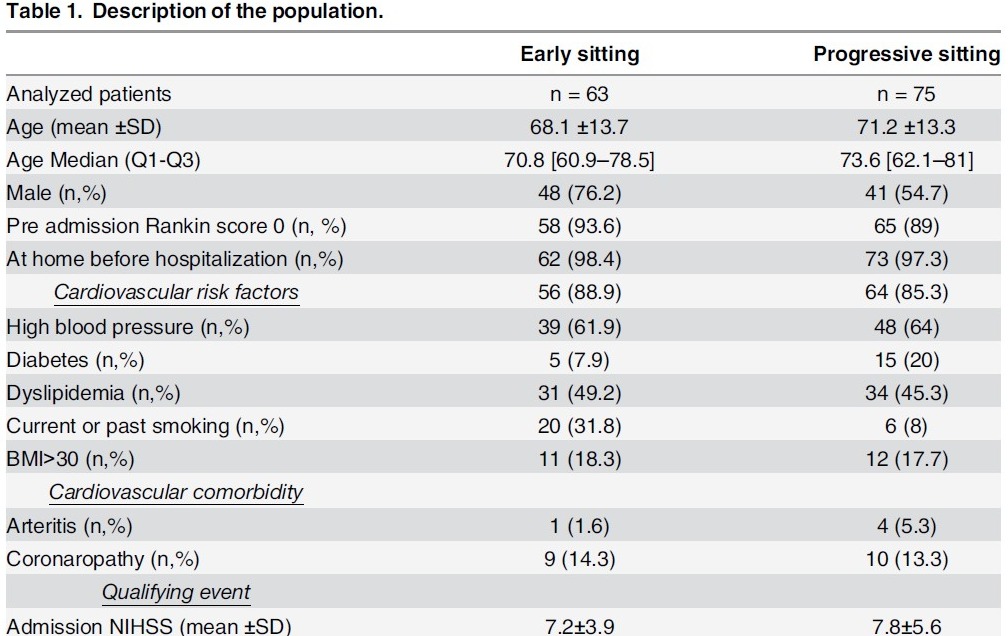
Fig.3整理和分析後形成有意義的知識
概念與流程示意圖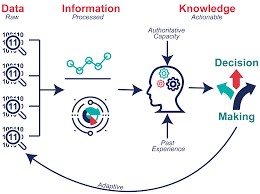
![]()
教育培訓
課程規劃核心為以「學習者」為中心進行「傳承」
以學習者為中心,結合陳秀敏博士十多年來的統計實務以及教學經驗,設計適合學員學習方式,開設課程,達到有效學習。
開設線上統計學院
SPSS基礎統計實戰班:第一次分析SCI研究就上手(上、下)
課程網址:https://medata.teaches.cc/
課程介紹1:https://www.youtube.com/watch?v=MPz2wqN0v2M
課程介紹2:https://www.youtube.com/watch?v=nd5A5duxO5E
臨床研究思維-Open your mind
課程網址:https://medata.teaches.cc/
課程介紹1:https://www.youtube.com/watch?v=yTHdBnCdSnY
課程介紹2 : https://www.youtube.com/watch?v=kE9tXraICqk
![]()
計畫撰寫與統計諮詢


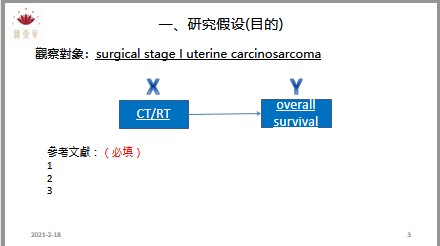
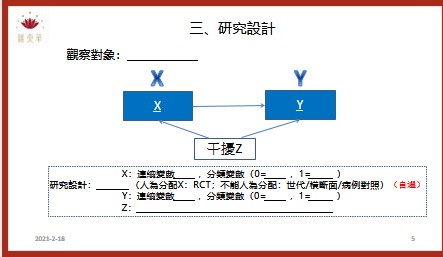
為了讓匯東華的顧客與學員有更好的合作和消費體驗,故匯東華特別依據營業項目開發周邊產品,提供使用、購買。目前已有針對公共衛生師的題庫以及模擬試題,未來將針對醫學研究領域發展產品。